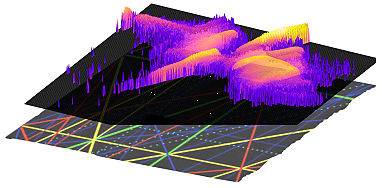
Chemistry is all about making and breaking of bonds and the rate at which they do so. To break a specific bond all that has to be done is to excite that bond and dump energy in excess of the bond strength. With some luck the deposited energy will stay put for a few vibrational time periods (about a few hundred femtoseconds) and then the bond snaps. Turns out that this viewpoint is far too naive due to the fact that molecules excited to such high energies have complicated intramolecular dynamics. The excited mode is coupled to many other modes and thus the initially localized energy flows rapidly into many other, perhaps undesirable, modes. In other words, the molecular choreography is very complicated. Sometimes it is so complicated that it is simple! This flow of energy within a molecule is the phenomenon of Intramolecular Vibrational energy Redistribution (IVR). The questions that we are, as many other chemical physicists in the world are, interested in: Where does the energy flow? How? Why? How fast? How is this classical notion of ball-and-spring vibrational motion encoded in the quantum eigenstates? Explaining and hence understanding this molecular choreography will let us control molecular reaction dynamics.
Our group is working on unraveling the IVR pathways in molecules from classical, semiclassical and quantum viewpoints. IVR is facilitated in a molecule by chains of nonlinear resonances which form a intricate network - sort of a transport network complete with highways, by-lanes and dead-ends. What part of this network is utilized by the classical dynamics? An important question is whether the quantum dynamics respects the classical resonance network or does it use a shortcut, known as dynamical tunneling, to give rise to novel quantum IVR pathways. Perhaps, a detailed knowledge of this resonance road map will allow us to shut down some of the highways leading to controlled IVR and thus give mode-specific chemistry a fair chance to happen. Amongst other things, our research sheds new light on the mechanism of coherent control of gas phase reaction dynamics.