|
Atomic Absorption Spectrometer |
||||
|
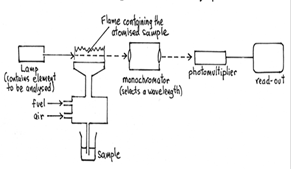
The Atomic absorption (AA) spectrometer is used to analyze metals at very low concentrations, typically in the parts per million (ppm) or parts per billion (ppb) ranges. A liquid sample containing dissolved material whose concentration is to be measured is aspirated into a thin, wide AA flame, or is introduced into a small carbon furnace which is heated to a high temperature. The Atomic absorption (AA) spectrometer (Varian AA240) (Figure 1) was established in the Core Lab (201D) in 2007 with the DRDO grant of the Institute. Basic Principle: AAS is the measurement of absorption of radiation by free atoms. The total amount of absorption depends on the number of free atoms present and the degree to which the free atoms absorb the radiation. At the high temperature of the AA flame, which may be either oxy-acetylene as used here, or nitrous oxide/acetylene, the sample is broken down into atoms and it is the concentration of these atoms that is measured (Figure 2). Unique Features: Fast Sequential capabilities with four lamps, so we can determine more elements in rapid sequence from one sample analysis. Fully automatic wavelength and slit selection simplifies operation, even for novice users. Completely sealed optics with quartz overcoated mirrors offer protection in dusty or corrosive environment. The air purge system is fitted inside the instrument eliminating the chance of corrosion in rugged or corrosive environments. |
||||
|
Location: |
||||
|
Department of Chemical Engineering,Core Lab 201D, |
||||
|
Contact: |
||||
|
Prof. Nishith Verma |
||||